A good habituation is a key success factor with DECRO® solution

DECRO® is the first non-invasive cardiorespiratory monitoring solution using a connected jacket for small laboratory animals. The use of DECRO allows to refine the protocols and reduce the number of animals used in a study.
The solution allows the acquisition of data in a vigile animal, socialized with its congeners, and free to move under different experimental conditions (Figure 1).
1. Habituation
In your animal studies, some procedures will require habituation. This step will decrease the effect of an external stimulus on an individual by repeatedly exposing them to it (Grissom and Bhatnagar 2009).
On the same principle, a habituation to the DECRO® jacket will be necessary before starting a study. The rest of this article describes the procedure recommended by ETISENSE. This is based on several studies carried out in rats (Wistar and Sprague Dawley), in different environments and animal houses.

Figure 1: Two rats socialized and equipped with DECRO®
This procedure (Figure 2) remains adjustable and will be adapted to your needs, to your experimental constraints and to the total duration for which the animals will be equipped. The habituation generally takes place over three days in a progressive manner in terms of the number and duration of exposure of the different “new” elements that are presented to the animals (manipulator, environment, jacket…).
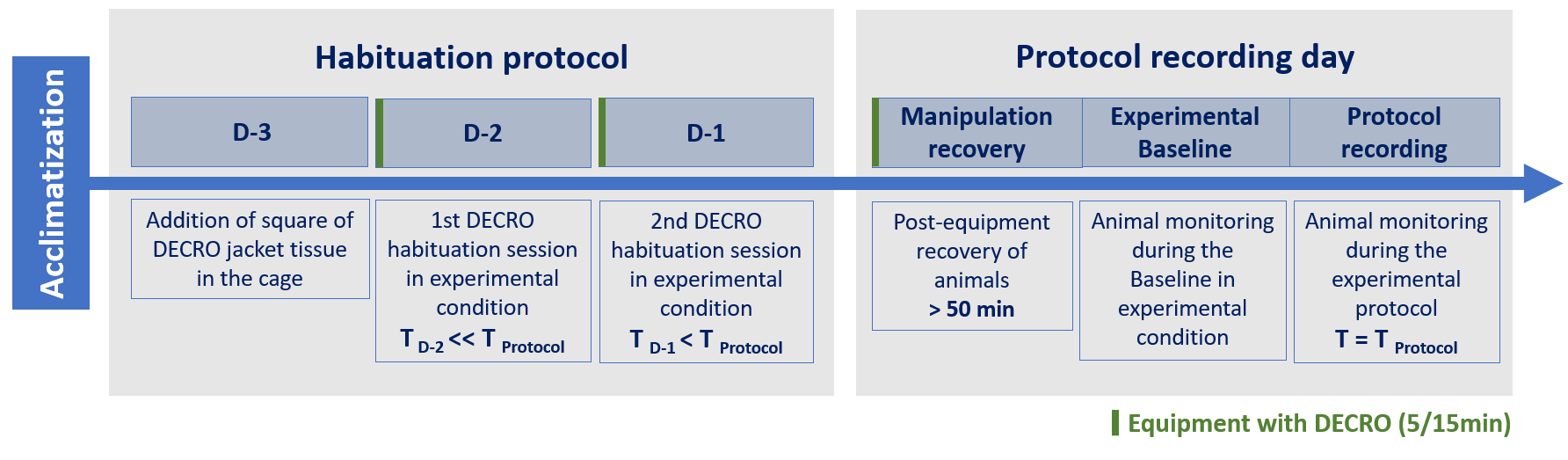
Figure 2: DECRO® habituation protocol
First, after the acclimatization phase in the animal house, we advise you to place the fabric square of the jacket provided with the system in the housing cage 24 hours before the first equipment in order to desensitize the animals to the novelty of the material. This step is strongly recommended for protocols in socialization or with a wearing time superior to 12 hours.
The animals can then be habituated to the DECRO® solution equipping procedure (Fares et al. 2020). We advise performing two equipment’s sessions prior to the protocol. Depending on the total equipment time desired during the protocol, the time of the first equipment can vary between 1 and 5 hours. The time for the second equipment will be longer (up to 8 hours). These first set-up sessions can also allow you to make “test” recordings and to familiarize yourself with the entire solution (software, control unit, transmitter…).
2. Recovery and Baseline
Any manipulation performed in the animal, even that qualified as light (e.g., restraint, injection…) can cause stress in the animal (Meijer et al. 2006). Even after repeated sessions, the restraint performed to equip the animals can generate variations in physiological parameters (Figure 3 (Flénet et al. 2019)).
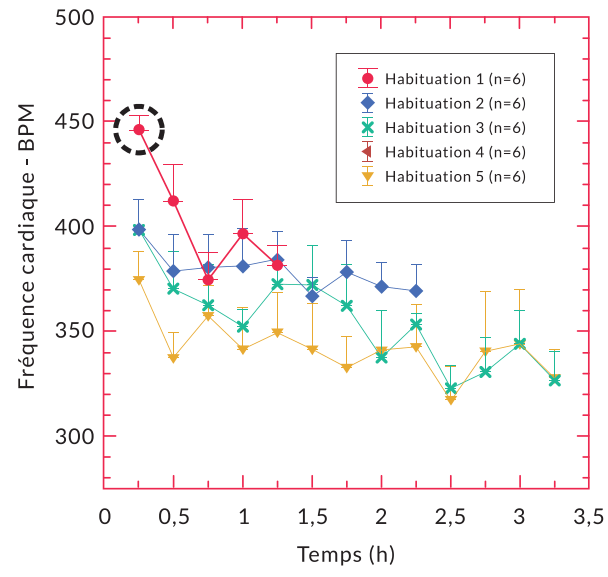
Figure 3 : Changes in heart rate over time as a function of the number of DECRO® habituation sessions performed on 6 Sprague Dawley rats (Flénet et al. 2019).
On recording days, we recommend a minimum acclimation period of 50 minutes to recover from the manipulation. The recommended acclimation time was based on observations of heart rate recovery in different environments showing recovery in 45 to 50 min. This is consistent with other manipulations performed in rodents (Meijer et al. 2006).
This should be followed by a 30 min to 1 h baseline if you need to record a baseline state of the animal. If you can, we advise you to check the heart and breathing rate of your animals to make sure of it before continuing the protocol.
Finally, if the habituation session could not be performed, the acclimatization period on the day of recording should be extended. In this case we recommend a minimum acclimatization period of two hours.
If you would like more information about the solution or if you would like to get advice on your experimental design with DECRO®, contact us.
References
Fares R., Boire A., Eynard C., Flenet T. Simultaneous non-invasive telemetric electrocardiogram and respiratory measurement with a connected jacket (DECRO system) in rats [Internet]. Protocol Exchange; 2020 May. Available from: https://protocolexchange.researchsquare.com/article/pex-860/v1
Flénet F., CHASTEL E., EYNARD E., MOMTAZ A., BOIXEL C., BARRET H. Raffinement du suivi cardiorespiratoire chez le rongeur : développement et validation d’une nouvelle solution de télémétrie. Revue des Sciences et Techniques de l’Animal de Laboratoire AFSTAL. 2019 me trim;Volume 47:14–20.
Grissom N., Bhatnagar S.. Habituation to repeated stress: Get used to it. Neurobiology of Learning and Memory. 2009 Sep;92(2):215–24.
Meijer MK., Spruijt BM., van Zutphen LFM., Baumans V. Effect of restraint and injection methods on heart rate and body temperature in mice. Lab Anim. 2006 Oct 1;40(4):382–91.
